Charles River Laboratories International, Inc. CRL inked an AAV9 production program deal with Navega Therapeutics. The deal will grant Navega Therapeutics access to Charles River’s contract development and manufacturing capabilities and advisory services to produce the AAV gene therapy candidate NT-Z001 for phase 1 clinical trials.
The recent move will bolster Charles River’s Biologics Solutions.
More on the News
Using the accuracy of its AI-enabled zinc finger epigenome regulatory technology, Navega Therapeutics is investigating a therapy approach for chronic pain associated with rare disorders such as tiny fiber neuropathy and primary erythromelalgia.
The non-opioid gene therapy developed by Navega Therapeutics for chronic pain may potentially be utilized to treat neuropathic and inflammatory pain, two more intractable pain indications, as more than 17 million U.S. citizens suffer from high-impact chronic pain.
Strategic Implications
The partnership with Navega Therapeutics will enable Charles River to leverage its strong AAV portfolio and leading gene therapy CDMO capabilities. The company is optimistic that patients with chronic pain will have access to Navega Therapeutics’ gene therapy.
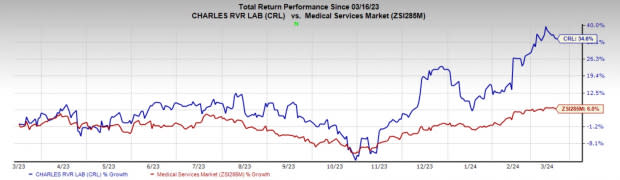
Image Source: Zacks Investment Research
Through collaboration, Navega Therapeutics intends to utilize Charles River's pre-made plasmid products, custom plasmid synthesis capabilities and Good Manufacturing Practice (GMP)-grade AAV production to get NT-Z001 to the clinic.
Industry Prospects
Per a report by Coherent Market Insights, the global cell and gene therapy market was valued at $22.7 billion in 2023 and is anticipated to witness a CAGR of 28.7% between 2023 and 2030. Factors like the increasing demand for innovative treatments and the developing interest in cell and gene treatments for cancer treatments are likely to drive the market.
Progress in Cell and Gene Therapy
Over the years, Charles River significantly enhanced its cell and gene portfolio through a mix of acquisitions, capacity expansions and manufacturing platform launches, including eXpDNA for plasmids, nAAVigation for AAV and Lentivation for lentiviral vectors.
In January 2024, Charles River launched its off-the-shelf Rep/Cap plasmid offering. Per the company, it has been designed to streamline adeno-associated virus (AAV)-based gene therapy programs.
In December 2023, Charles River achieved an important milestone in its strategic collaboration with Vertex Pharmaceuticals to manufacture CASGEVY (exagamglogene autotemcel [exa-cel]). The company’s Memphis facility was approved to manufacture Vertex’s CASGEVY — the first-ever gene-edited therapy in the world that targets severe sickle cell disease.