
allanswart
allanswart
Vistagen Therapeutics (NASDAQ:VTGN) has proprietary platform technologies to discover and develop drugs that could redefine the treatment of mental health disorders. With a potential NDA-enabling Phase III program already funded, Vistagen is approaching some significant milestones that could be potent catalysts for the ticker. Moreover, the company’s deep pipeline of neuroscience candidates, each targeting individual disorders with inventive mechanisms of action, distinguishes their approach to contemporary standards of care. The company's portfolio offers investors the rare opportunity to tap into massive markets of social anxiety disorder and major depressive disorder, at a $110M market cap. I believe VTGN offers an enticing risk-reward at this time and will be on my watch list going into Q2.
I intend to provide a brief background on Vistagen and its recent performance. Then, I will present some potential catalysts related to Fasedienol as well as downside risks. Finally, I discuss my strategy for VTGN.
Vistagen is a biopharma developing treatments for psychiatric and neurological ailments including anxiety, depression, and related disorders. The company’s clinical-stage pipeline comprises five innovative pherine-based candidates that target chemosensory neurons in the nasal passages without systemic absorption or brain receptor binding. These candidates promise to influence core neural circuitry directly. In addition, the company has an oral prodrug candidate that aims to modulate NMDA receptor activity without complete blockade.
The company’s lead program, Fasedienol (PH94B), is a neuroactive nasal spray that could potentially be a breakthrough in the management of social anxiety disorder (SAD). Fasedienol’s Phase III trial results showcased its ability to rapidly improve symptoms of anxiety after one dose, along with a safety profile and potential for fast-acting relief. At the moment, Vistagen is preparing to initiate their PALISADE Phase III program with PALISADE-3 and PALISADE-4 studies that are slated to commence in 2024. If successful, these could be sufficient to file for regulatory approval.
Vistagen’s Itruvone (PH10) is being tested for Major Depressive Disorder (MDD), without the risk of common side effects linked to existing antidepressants. The company’s Phase II trials established significant symptom reduction compared to placebo, positioning it as a potential generational treatment for depression. Following the successful Phase IIA trial in MDD, Vistagen is gearing up for a Phase IIB study.
The company is taking on vasomotor symptoms and PMDD, with PH80, which has shown statistically significant marks indicating its potential to address unmet needs in these areas. Addressing vasomotor symptoms (VMS) due to menopause, PH80 offers a fresh approach with rapid relief similar to a rescue inhaler. PH80’s Phase IIA data highlighted its efficacy in reducing hot flashes and improving quality of life for menopausal women.
Meanwhile, AV-101, an oral NMDA receptor antagonist, is being developed as a versatile therapeutic asset with implications for MDD and neuropathic pain. However, with ongoing clinical evaluations demonstrating its efficacy and safety profile, Vistagen believes AV-101 could be an “alternative for multiple CNS disorders.” The company is looking into moving AV-101 forward into Phase IIA studies either on their own, or with collaborators.
PH15 is another Vistagen pherine nasal spray candidate used for cognitive impairment induced by mental fatigue and potentially other conditions. Like the company’s other pherine-based nasal sprays, PH15 is promoted to have a rapid-onset mechanism of action, distinctive from existing treatments. Currently, the company is working on a path forward for a Phase II study and is weighing the possibility of seeking collaborators.
PH284 is another one of Vistagen’s pherine nasal sprays targeting cachexia. With its rapid-onset MOA, PH284 offers a promising avenue by enhancing the patient’s feelings of hunger, potentially leading to improved nutritional intake and weight management in patients grappling with cachexia and evaluating the studies necessary to advance its clinical development. The next step is moving PH284 into Phase II, either independently or in collaboration with a partner.
Since mid-2021, Vistagen has recorded a negative net income, with fluctuations ranging from -$6.3M to -$19.8M, with their highest negative free cash flow of -$11M in Q3 of 2022. To fund the company, the company has executed some notable financing activities in calendar Q3 2023 of $34.2M and another $93.5M in the following quarter.
As of their last earnings report, Vistagen has maintained a robust cash position, with $126.6M in cash and cash equivalents at the end of 2023. In addition, the company recently filed for a $350M mixed-shelf offering. This strong financial position gives Vistagen ample liquidity to support its ongoing operations, chase strategic initiatives, and fund its clinical development programs.
As Vistagen continues to advance its pipeline and solidify its position as a leader in mental health therapeutics, investors must remain vigilant about some key upcoming catalysts. One of Vistagen's key catalysts comes from positive clinical trial results, particularly for Fasedienol. Fasedienol is heading into the PALISADE Phase III program with PALISADE-3 and PALISADE-4 studies, which could be ready for readouts in 2025. It is possible that one of these readouts could be sufficient to apply for FDA approval, thus, making them powerful catalysts that could improve the company's valuation and attract additional investor interest.
If approved, Fasedienol would not only offer a potential game-changer that could become a blockbuster drug but could validate the company’s pherine nasal technology that makes up the majority of their mental health pipeline. Keep in mind, Fasedienol would be the first FDA-approved therapy for acute-SAD… So, Fasedienol could be used by SAD patients as a rescue inhaler by asthmatics. Again, at the moment, SAD patients do not have anything that they can self-administer that provides them with this level of relief.
Vistagen FDA-Approved Drug Profiles For SAD (Vistagen)
Furthermore, not only would the company become a contender in the SAD market, but a potential contender in the mental health therapeutics market, which is gigantic and expanding. Itruvone could have the same impact in on MDD, which alone is affecting over 264 million individuals around the globe and causing significant socioeconomic burdens. Therefore, Vistagen's pipeline positions them as a potential market disruptor that could address the unmet needs of millions of patients, all of which could be partially validated by Fasedienol’s success.
Another potential scenario that could be unlocked from a positive Fasedienol readout would be interested in strategic partnerships and collaborations. The company has mentioned that they are considering finding partnerships for some of their other pipeline programs. Furthermore, the company might attract a commercial partner for Fasedienol, which would likely include an upfront payment, milestones, and royalties.
Vistagen has plenty of challenges and uncertainties that investors ought to consider when managing their position. Although Fasedienol’s data thus far has been encouraging, the path to approval still has plenty of hurdles. Clinical development is inherently risky, the readout from the PALISADE-3 and PALISADE-4 studies will be critical inflection points for Vistagen's trajectory. So, a negative readout from these studies could demolish the share price. Fasedienol’s data will likely have to verify that it differentiates from contemporary products to fend off a potential sell-off.
Another risk to consider is commercialization. At this time, I don’t see any plans to get Fasedienol on the market. Vistagen might be considering a commercial partner in the United States, which typically bodes well for smaller outfits. However, I would suspect the Street would be a bit apprehensive if the company does decide to “go-it-alone” in commercialization, due to the need to build a commercial team and a significant bump in OpEx. Even if the company were to assemble this infrastructure, the Street would likely question their ability to be competitive against numerous companies vying for market share, including several Big Pharma companies.
Lastly, investors need to keep an eye on the financials. At the moment, the company has a healthy $126.6M cash position. On the other hand, we could see a bump in expenses as the company progresses their pipeline and moves Fasedienol closer to the finish line. A setback in the regulatory process and failure in the clinic will most likely require the company to prolong their cash burn. On the other hand, an FDA approval could result in additional spending to build a commercial team, manufacturing, marketing, sales, etc.
Indeed, I am still bullish on VTGN, however, additional risks arise with clinical success. As a result, I am giving VGTN a conviction level of 3 out of 5 and would be placed in my Compounding Healthcare “Bio Boom” Speculative Portfolio.
I believe VTGN offers an enticing risk-reward at this time considering Fasedienol has the potential to be a blockbuster drug, and yet, the ticker is trading with a negative enterprise value.
VTGN Capital Structure (Seeking Alpha)
It appears as if VTGN is flying under the radar amongst retail investors considering the ticker only has roughly 5.41 followers on Seeking Alpha. In addition, the Street is not doing a great job covering the company with only four analysts forecasting earnings and only one analyst brave enough to present their views into the next decade.
Vistagen Analyst Annual Revenue Estimates (Seeking Alpha)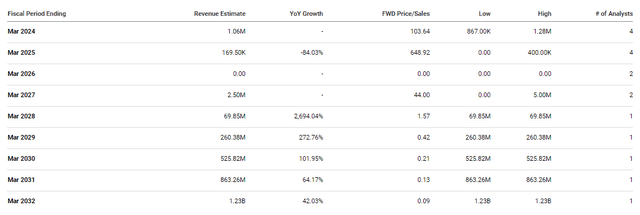
Still, that one analyst does project Vistagen going from reporting a minuscule amount of revenue, to crossing blockbuster status in 2032. Assuming the analyst is correct, Vistagen would pull in $1.23B in 2032, which would be a forward price-to-sales of 0.09x. The industry’s average price-to-sales is roughly 4x, so $1.23B would be roughly $182 per share. Of course, the company will most likely have to execute some dilutive financing, which would cut that target down dramatically. Unfortunately, we don’t have a clear view of their expenses over the next several years. In addition, it is possible they might find a commercial partner to split the revenue with, which would alter these estimates. Still, these revenue projections illustrate an attractive risk-reward for VTGN at these prices.
Vistagen Therapeutics represents a compelling investment opportunity in the burgeoning field of mental health therapeutics. With an innovative platform technology and a robust pipeline, Vistagen is well-positioned to capitalize on the growing demand for novel treatments in psychiatry and neurology. However, I will remain vigilant about Vistagen's financials and long-term plans for commercialization.
Back in August of 2023, VTGN spiked up following their Phase III data, but has under controlled selling pressure that is being piloted by downtrend rays and anchored-VWAP. So, my initial plan of attack would be wait for VTGN to break above the downtrend ray and the proximal anchored-VWAP, which would be around $4.76 per share.
VTGN Daily Chart (Trendspider)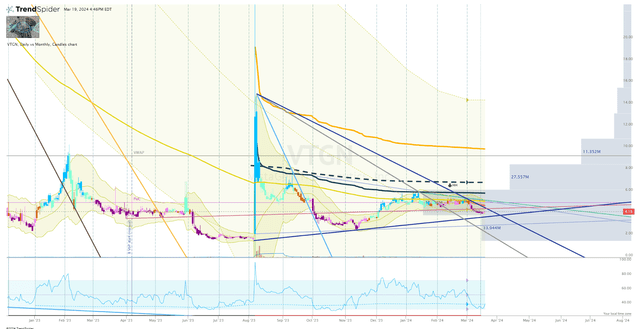
However, if the share price fails to break the downtrend ray and drops below the uptrend, I will consider establishing a position if the ticker can bounce off the lows ($1.40) from back in August. Once I have established a position, I will look to place a buy order following updates or earnings reports.