Design Cells
Design Cells
United Therapeutics Corporation (NASDAQ:UTHR) is a biotechnology and pharmaceutical company specializing in cardiovascular disorder treatments such as Pulmonary Arterial Hypertension [PAH], Pulmonary Hypertension associated with Interstitial Lung Disease [PH-ILD], and pediatric oncology drugs. Its research pipeline advances its lung disorders platforms, develops novel biologics to treat sepsis, and generates various organ manufacturing programs that are transforming the landscape of organ transplantation with innovative new systems. Unfortunately for UTHR, its main revenue source (Tyvaso) is legally challenged, which endangers its future growth rate. As such, this could become a significant valuation headwind for the stock, which I don't think investors should ignore. Hence, I lean neutral on the stock and rate it a "hold" for now.
United Therapeutics is a biotechnology and pharmaceutical company founded in 1996. It is headquartered in Silver Spring, Maryland, and Research Triangle Park, North Carolina, with the legal and creative departments in Washington, D.C. UTHR has additional facilities in Bromont, Quebec; Melbourne and Jacksonville, Florida; and Manchester, New Hampshire. UTHR specializes in organ manufacturing for transplantation and developing and commercializing drugs for cardiovascular disorders. UTHR generates revenues mainly by commercializing pharmaceuticals and medical devices for treating PAH and related cardiovascular disorders.
Source: United Therapeutics Corporation Fourth Quarter 2023 Financial Update. February 21, 2024.
Let's look a bit deeper into the company's IP portfolio. UTHR has a multifarious product portfolio of therapies for vascular conditions such as Pulmonary Arterial Hypertension [PAH] and Pulmonary hypertension associated with Interstitial Lung Disease [PH-ILD]. UTHR has extended its offerings into pediatric oncology with high-risk neuroblastoma therapy. The company has five FDA-approved medications to treat these vascular diseases: Orenitram, Tyvaso, Tyvaso DPI, Remodulin, and Adcirca (tadalafil). UTHR also offers an FDA-approved drug called Unituxin (dinutuximab), a monoclonal antibody for neuroblastoma treatment.
Moreover, UTHR's pipeline consists of various programs in organ manufacturing, novel biologics, Tyvaso, and Remodulin platforms. Teton (inhaled treprostinil) for idiopathic pulmonary fibrosis [IPF] and Advance (IP receptor agonist) for PAH are in phase 3 of the clinical trials. RemoPro (injected treprostinil prodrug) indicated for PAH is in phase 2.
Another promising R&D program is UTHR's genetically enhanced Mesenchymal stem cells [GEMS] study in phase 2. This study explores the potential use of stem cells with augmented regenerative capabilities to rejuvenate damaged tissues. UTHR's GEMS technology could be revolutionary for treating sepsis, a severe response to infection that provokes inflammation and can lead to organ failure and death as organs are damaged. Moreover, sepsis has a high mortality rate, so UTHR's technology is invaluable in some cases, and further advancements in this area could unlock additional shareholder value as they progress. For context, sepsis affects approximately 1.7 million adults in the US annually so this underserved population could become one of UTHR's value drivers in the future.
Source: United Therapeutics Corporation Fourth Quarter 2023 Financial Update. February 21, 2024.
Furthermore, UTHR also has programs as part of its organ manufacturing platform in the preclinical stage. These studies include Kidney (xenokidney), ThymoKidney (xenokidney), Uheart (xenoheart), ULobe (Lung Lobes), and ULung (3DAP Lungs). These studies involve xenotransplantation using modified animal organs with a combination of bioengineering techniques, such as 3D printing technologies, to create functioning organs for transplantation to address the shortage of human organ donation to save the lives of patients with severe organ failure.
Source: United Therapeutics Corporation Fourth Quarter 2023 Financial Update. February 21, 2024.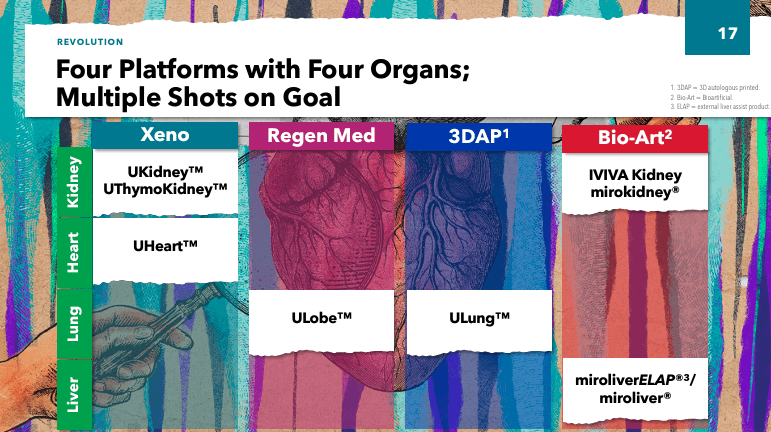
UTHR also conducts clinical trials for centralized ex-vivo lung perfusion service devices under the EVLP (ex-vivo lung perfusion) system. This instrument can repair lungs previously considered unsuitable for transplantation by circulating a nutrient-rich solution outside the body. Thus, this intervention could increase the number of available lungs for transplants and save lives. Long wait times for lung transplants can cost lives as patient conditions worsen quickly without them. According to the UNOS, the gap between patients waiting for lung transplants and available organs has widened in recent years, showcasing a timely market potential for UTHR if these clinical trials are successful.
During the company's latest earnings call, UTHR's executives celebrated another consecutive quarter of record revenue with over 20% growth for Q4 2023 and favorable full-year 2023 results. Additionally, management announced a royalty transaction related to their Tyvaso Dry Powder Inhaler [DPI] product, executed by UTHR's partner MannKind Corporation that sold a tenth of their royalty stream to Sagard Healthcare Partners for $150 million for a 1% percent royalty stream, plus additional potential milestone payments. UTHR's executives highlighted that this value could imply that 100% of Tyvaso's DPI revenue is valued at $15 billion for one of its products, which is quite higher than the current market cap of UTHR and implies a significant value driver that doesn't seem to be reflected in its market cap today.
Source: United Therapeutics Corporation Fourth Quarter 2023 Financial Update. February 21, 2024.
Additionally, UTHR's management outlined their strategy based on 1) leveraging the existing business, 2) advancing the near-term pipeline, and 3) pursuing long-term growth by developing manufactured organ technologies. This is a mature company, so it's not the typical small biotech stock with high-risk and high-reward potential but rather a relatively stable business in the sector. As such, executives provided updates in xenotransplantation and regenerative medicine, such as the IND issued by the FDA for the Miroliver External Liver-Assist Product [ELAP] program to create livers using tissue engineering, cell biology and biomaterial techniques. The primary function of the MiroliverELAP system is to externally support the liver functions of patients with acute liver failure to reduce the need for transplantation, giving time for the patient's liver to heal.
Overall, I believe that UTHR's product portfolio is robust, with ongoing promising clinical trials in its pipeline. Thus, I think it's safe to consider UTHR well-positioned for sustained growth due to its strategy of leveraging its commercial products while investing in innovative research and groundbreaking projects.
In this positive scenario, a storm looms over one of the company's products when a US appeals court rejected UTHR's attempt to revive a patent over Tyvaso that an administrative tribunal canceled. The Patent Trial and Appeal Board concluded that two scientific papers' abstracts "were sufficiently disseminated and each constituted a printed publication." The ruling affirmed the cancellation of the patent. This move will get Liquidia, a smaller competitor, closer to obtaining FDA approval for its Yutrepia drug, like Tyvaso. This situation is a significant challenge for UTHR because it impacts the market exclusivity over Tyvaso, a key product for the company.
Source: UTHR's 2023 10-K report.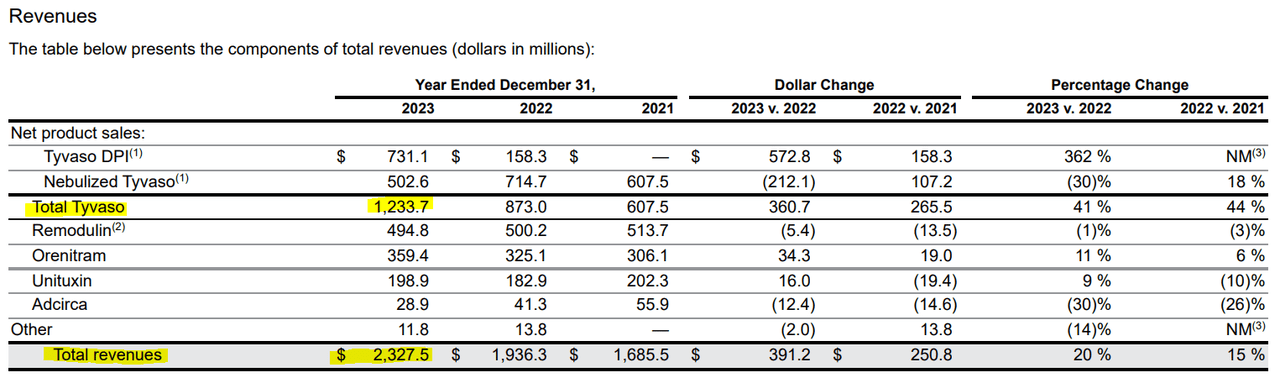
For context, UTHR's total revenues for 2023 were $2.33 billion, of which 53.0% came from Tyvaso's IP. This means that if Liquidia is successful in its legal challenge, then a significant portion of UTHR's revenues will be directly challenged. This is a substantial risk to the stock's investment thesis, and given that it already trades at a relatively high P/S ratio of 4.7, it could become a significant valuation headwind going forward if revenue growth slows down due to increased competition. For context, the sector median P/S multiple is 3.8, which suggests that UTHR trades at a premium relative to peers. This tempers my optimism about the stock itself, even though I remain optimistic about its underlying business. The good news is that UTHR's balance sheet is strong, with roughly $3.00 billion in cash and equivalents against $732.8 million in total debt. So, the company has ample resources to fund its legal battle, finance ongoing operations, and invest in its R&D programs.
Consequently, as of February 2024, UTHR continues litigation against the FDA for handling the Liquidia amendment to a new drug application [NDA] associated with PH_ILD of its proposed inhaled dry powder treprostinil product. UTRH alleges that Liquidia should seek approval for this second indication by filing a new NDA to ensure proper review of new drug applications. This legal action aims to protect UTHR's market interest and ensure that competitors adhere to regulatory standards, potentially seeking to delay Liquidia's product entry into the market. Yet, from an investment perspective, I believe that at this point, UTHR's short-term narrative will mostly focus on legal matters. If UTHR's challenge is successful, this would lift a significant risk and should become a good "buy" for long-term investors. But for now, I accept this legal uncertainty is too substantial to ignore, which is why I lean neutral on UTHR for the time being.
UTHR's stock has been treading water for the last year. (Source: TradingView.)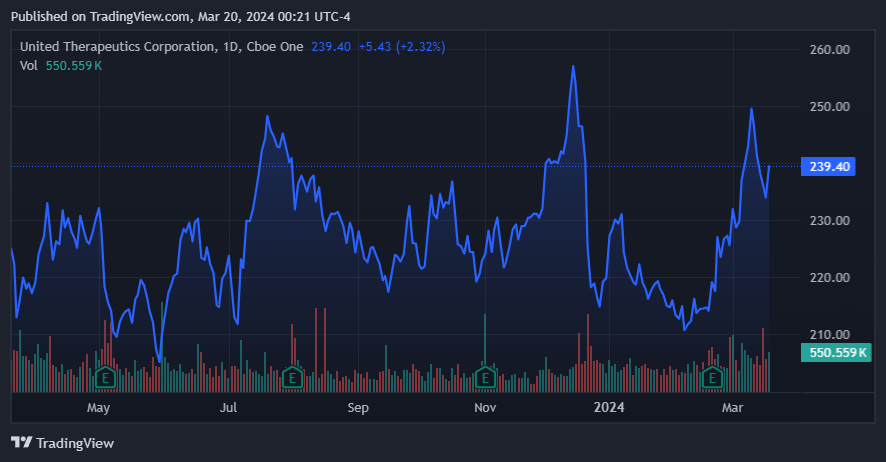
Overall, UTHR has exceedingly promising IP and seems quite profitable while continuing to develop its upcoming product pipeline through clinical trials. Yet for now, roughly half of UTHR's revenues come from its Tyvaso IP, also facing potentially increased competitive pressure. This ongoing legal challenge could set the tone for the company's long-term revenue growth rate and, by extension, its appropriate valuation multiple. Since UTHR already appears to trade at a premium relative to peers, I believe it's prudent to acknowledge legal risks affecting its main revenue source. Thus, despite the promising fundamentals, I remain neutral on the stock until this legal dispute is settled.