Shares of Beam Therapeutics Inc. BEAM have rallied 27.3% in the past three months compared with the industry’s increase of 9.8%.
Beam’s pipeline of gene editing therapies is based on its proprietary base editing technology. The company is advancing its base editing technology across three disease area portfolios, namely, hematology, immunology/oncology and genetic diseases.
The company is developing its leading ex-vivo genome-editing candidate, BEAM-101, for the treatment of sickle cell disease (SCD). The candidate is currently being evaluated in the phase I/II BEACON study for treating SCD. Initial data from the study is expected in 2024. Steady progress in the development of BEAM-101 could be a catalyst for the stock to increase during the abovementioned time period.
Also, the FDA recently approved two gene therapies for treating SCD. This has put the spotlight on companies that have promising gene editing candidates in their pipeline, including BEAM.
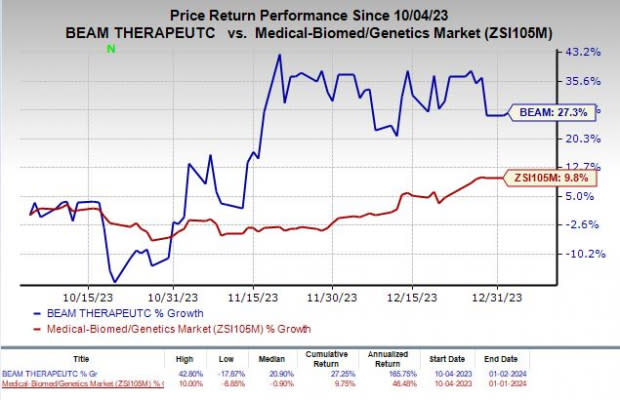
Image Source: Zacks Investment Research
We note that in October 2023, Beam announced a strategic restructuring to streamline its current portfolio of pipeline candidates and business operations. Following the portfolio prioritization, BEAM will mainly focus on the development of BEAM-101.
As part of the reprioritization, BEAM is looking to reduce its current headcount by 20%, which is likely to reduce costs.
Importantly, the restructuring and reprioritization efforts are expected to reduce the company’s operating expenses and extend the cash runway into 2026.
This apart, Beam is also developing in vivo base editor BEAM-302 for the treatment of alpha-1 antitrypsin deficiency. Regulatory applications to begin clinical studies on BEAM-302 in ex-U.S. markets are expected to be filed in the first quarter of 2024. The company will file an investigational new drug (IND) application with the FDA to begin clinical studies on BEAM-302 in the United States soon after.
The company is also planning to initiate a clinical study on BEAM-301 for the treatment of glycogen storage disease 1a in the United States. An IND application for BEAM-301 is expected to be filed in the first half of 2024.
Beam is evaluating BEAM-201 in a phase I/II study for treating patients with T-cell leukemia and T-cell lymphoblastic lymphoma and potentially other CD7+ malignancies.
Though still in the early stage, Beam’s pipeline holds great potential. If successfully developed and commercialized, BEAM-101 can significantly boost the company’s prospects and drive the stock in the days ahead.
However, competition in the target market remains a concern. A few other companies are also developing their own in-vivo and ex-vivo gene-editing therapies utilizing CRISPR/Cas9 nuclease technology.