Amicus Therapeutics, Inc.’s FOLD lead drug, Galafold (migalastat), is the first and only approved oral medicine for patients living with Fabry disease, having amenable genetic variants. The drug has shown solid uptake since its launch.
Sales of Galafold have been rising consistently over the past years. In the first nine months of 2023, the drug generated sales worth $281.2 million, increasing 16.6% on a year-over-year basis. Galafold is currently approved in several countries, including the United States, European Union (EU), United Kingdom and Japan.
In 2021, Galafold received label expansion in the EU for long-term treatment of adolescents aged 12 years and older with Fabry disease. Such label expansion, along with approvals in additional geographies, is likely to drive sales of the drug further in 2024 and beyond.
Shares of Amicus have gained 18.9% in the past year against the industry’s decline of 14.1%.
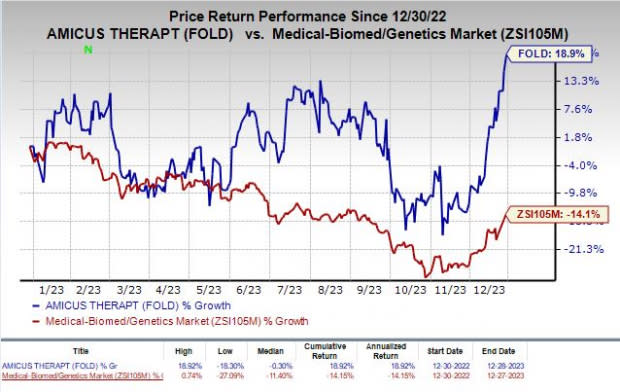
Image Source: Zacks Investment Research
In September 2023, the FDA approved Pombiliti (cipaglucosidase alfa) + Opfolda (miglustat) 65mg capsules, a two-component therapy for treating late-onset Pompe disease (LOPD) in adults, who weigh more than 40 kg and are not improving on their current enzyme replacement therapy.
The FDA nod was based on positive data from the phase III PROPEL study, the only randomized and controlled trial in LOPD.
Earlier in 2023, Pombiliti + Opfolda was approved in Europe, including the United Kingdom, for treating LOPD in adults. Global launches of Pombiliti and Opfolda are currently underway, including the United States, EU and United Kingdom.
The recent approval for Pombiliti + Opfolda in the United States is a huge boost and provides a significant commercial opportunity for Amicus in 2024 and beyond.
Though Amicus is riding on the success of Galafold, which drives the majority of the company’s top line, heavy dependence on the product for revenues remains a concern. Any regulatory setback for the drug will hurt the company’s growth prospects.
Also, several large pharmaceutical and biotechnology companies currently market and sell products for treating Fabry disease and Pompe disease. This acute competition remains a headwind for Amicus in the days ahead.
Amicus Therapeutics, Inc. Price and Consensus
Amicus Therapeutics, Inc. price-consensus-chart | Amicus Therapeutics, Inc. Quote
Zacks Rank & Stocks to Consider
Amicus currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the healthcare sector are Taro Pharmaceutical Industries Ltd. TARO, Entrada Therapeutics, Inc. TRDA and Puma Biotechnology, Inc. PBYI, each sporting a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.