Shares of Iovance Biotherapeutics, Inc. IOVA were down 18.7% on Dec 27 after the company announced a clinical update on its tumor infiltrating lymphocyte ("TIL") therapy, LN-145, in the non-small cell lung cancer (“NSCLC”) indication.
The phase II IOV-LUN-202 study is currently evaluating LN-145 for treating patients who have progressed on or after chemotherapy and anti-PD-1 therapy for advanced (unresectable/metastatic) NSCLC.
On Dec 22, 2023, the FDA placed a clinical hold on the IOV-LUN-202 study following a grade 5 (fatal) serious adverse effect, potentially related to the non-myeloablative lymphodepletion pre-conditioning regimen observed in the study.
Owing to the clinical hold, IOVA will pause the enrollment of new patients in the IOV-LUN-202 study. This is likely to delay the clinical advancement of LN-145 in the NSCLC indication.
Per management, patients who have already undergone tumor resection in the LN-145 TIL treatment regimen will continue to receive the same with extra precautions and risk mitigations.
Shares of Iovance have rallied 15.3% in the past year against the industry’s decline of 15%.
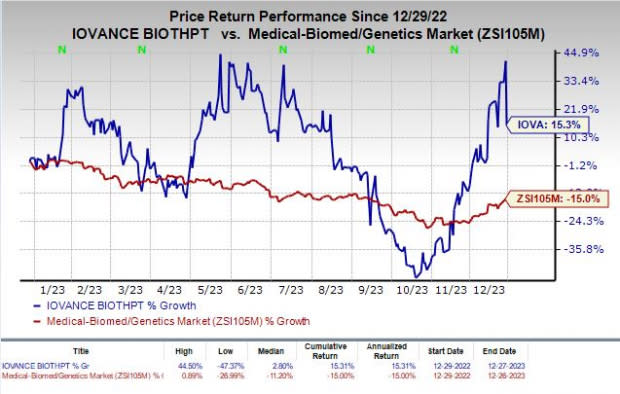
Image Source: Zacks Investment Research
Per the press latest release, the clinical hold on the IOV-LUN-202 study will not affect Iovance’s biologics license application (BLA) for the lead pipeline drug, lifileucel, in the advanced melanoma indication. The clinical hold will also not affect the company’s other ongoing clinical studies.
In May 2023, the FDA accepted IOVA’s BLA submission for lifileucel in melanoma. The BLA seeks accelerated approval for lifileucel to treat patients with advanced unresectable or metastatic melanoma, which progressed on or after prior anti-PD-1/L1 therapy.
With the FDA granting a priority review to the BLA, a decision from the regulatory body is expected on Feb 24, 2024.
In June 2023, Iovance randomized the first patient in the phase III TILVANCE-301 study, evaluating the combination of lifileucel and Merck’s blockbuster PD-L1 inhibitor, Keytruda, to treat immune checkpoint inhibitor naïve frontline metastatic melanoma. This late-stage study will also serve as a confirmatory study at the time of seeking full approval for the above BLA.
Regulatory filings for lifileucel in the melanoma indication in Canada and Europe are expected in 2024.
Zacks Rank & Stocks to Consider
Iovance currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks in the healthcare sector are Taro Pharmaceutical Industries Ltd. TARO, Entrada Therapeutics, Inc. TRDA and Puma Biotechnology, Inc. PBYI, each sporting a Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.