-
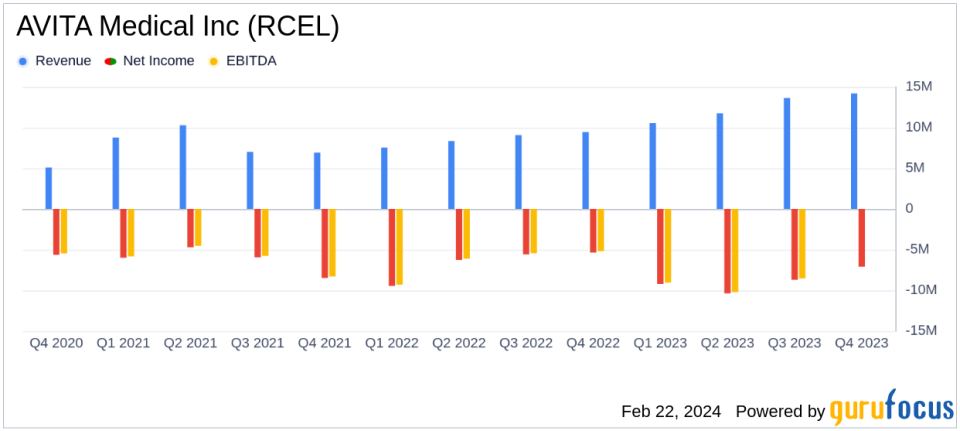
Q4 Commercial Revenue: Increased by approximately 50% to $14.1 million year-over-year.
-
Full-Year Commercial Revenue: Grew by approximately 46% to $49.8 million compared to 2022.
-
Gross Profit Margin: Improved to 87.3% in Q4 and 84.5% for the full year.
-
Net Loss: Q4 net loss widened to $7.1 million, full-year net loss increased to $35.4 million.
-
2024 Financial Guidance: Anticipates Q1 revenue between $14.8 to $15.6 million and full-year revenue between $78.5 to $84.5 million.
-
Cash Position: As of December 31, 2023, holds approximately $89.1 million in cash, cash equivalents, and marketable securities.
On February 22, 2024, AVITA Medical Inc (NASDAQ:RCEL) released its 8-K filing, detailing its financial results for the fourth quarter and full-year ended December 31, 2023. The company, known for its innovative RECELL systema device that creates Spray-on Skin Cells for treating burns and other skin defectshas reported significant revenue growth and provided optimistic financial guidance for 2024.
Company Overview
AVITA Medical is a regenerative medicine company with a primary focus on the U.S. market, where it is rolling out its RECELL system across approximately 136 burn centers. While the product is approved in various global markets, the company's current strategy is not to actively market outside the U.S. but to focus on the domestic region. Expansion plans are underway, with a launch expected in Japan through a distribution partner in the second half of fiscal 2022.
Financial Performance and Future Outlook
The company's financial highlights for the fourth quarter show a 50% increase in commercial revenue to $14.1 million, with a gross profit margin of 87.3%. For the full year, commercial revenue rose by 46% to $49.8 million, and the gross profit margin stood at 84.5%. AVITA Medical's cash reserves are robust, with approximately $89.1 million in cash, cash equivalents, and marketable securities as of year-end 2023.
CEO Jim Corbett commented on the company's progress, stating,
We ended the year with yet another quarter of significant growth, marking a year of extraordinary progress. In 2023, we successfully executed a series of strategic initiatives to transform our business."
These initiatives included expanding RECELL indications, doubling the commercial field organization, launching an expanded label for full-thickness skin defects, and establishing an international expansion plan.
Looking ahead, AVITA Medical plans to expand its field sales organization, integrate PermeaDerm Biosynthetic Wound Matrix into its portfolio, and expects FDA approval for RECELL GO with a commercial launch planned for May 31, 2024. The company also anticipates non-U.S. sales following the launch of RECELL in Germany, Austria, and Switzerland in January 2024.