Johnson & Johnson JNJ announced that the FDA granted full approval to its EFGR/MET inhibitor Rybrevant (amivantamab) in non-small cell lung cancer (NSCLC) indication.
Following the FDA decision, Rybrevant, in combination with chemotherapy (carboplatin-pemetrexed), is approved for the first-line treatment of patients with locally advanced or metastatic NSCLC with EGFR exon 20 insertion mutations. The drug was initially granted accelerated approval for a similar indication in May 2021.
Alongside the FDA approval, J&J announced that the National Comprehensive Cancer Network (NCCN) updated its guidelines recommending the Rybrevant-chemotherapy as a preferred first-line regimen for patients with NSCLC with EGFR exon 20 insertion mutations.
Both the approval and recommendation are based on positive top-line data from the phase III PAPILLON study, which evaluated Rybrevant plus chemotherapy in patients with newly-diagnosed advanced or metastatic NSCLC with EGFR exon 20 insertion mutations. Patients who received the Rybrevant-chemotherapy combination achieved a 61% reduction in the risk of disease progression or death compared to chemotherapy alone.
A regulatory filing is currently under review in the European Union seeking marketing approval/authorization for a similar indication, supported by data from the PAPILLON study. A final decision is expected in the coming months.
Shares of J&J have gained 3.4% year to date compared with the industry’s 13.6% growth.
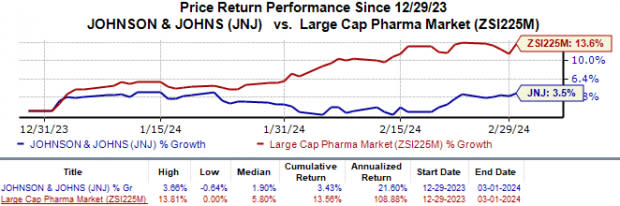
Image Source: Zacks Investment Research
During fourth-quarter 2023, J&J submitted two other supplemental biologics license application (sBLA) filings with the FDA seeking approval for combination therapies with Rybrevant in patients as first-line and second-line treatments for locally advanced or metastatic NSCLC with EGFR exon 19 deletions (ex19del) or L858R substitution mutations.
The first sBLA, filed in November, seeks the FDA’s approval for Rybrevant in combination with chemotherapy (carboplatin-pemetrexed) as a treatment for patients with locally advanced or metastatic NSCLC with EGFR ex19del or L858R substitution mutations whose disease has progressed on or after receiving AstraZeneca’s Tagrisso (osimertinib). This filing is supported by data from the late-stage MARIPOSA-2 study. A similar filing was submitted in the EU last November.
The second sBLA, filed in December, seeks the agency’s approval for Rybrevant plus oral EGFR-TKI inhibitor lazertinib for the first-line treatment of adult patients with locally advanced or metastatic NSCLC with EGFR ex19del or L858R substitution mutations. This submission is based on data from the phase III MARIPOSA study. A similar filing was also submitted in the EU last month.