Orchard Therapeutics plc ORTX, a rare disease gene therapy biotech, announced entering into a definitive agreement to be acquired by the Japanese pharmaceutical company, Kyowa Kirin Co., Ltd, for a consideration of $16 per American Depositary Share (ADS) in cash, which amounts to a total transaction value of approximately $387.4 million.
The total transaction value represents a premium of 144% to ORTX’s average price per ADS over the past 30 days ended Oct 4, 2023.
Per the transaction terms, Orchard’s shareholders will also receive an additional contingent value right (CVR) of $1 per ADS, upon the approval of Libmeldy (OTL-200) for the treatment of metachromatic leukodystrophy (MLD) in the United States.
Libmeldy is currently approved in the EU and U.K. to treat late infantile and early juvenile MLD patients. In the United States, Orchard has already filed for the regulatory approval of Libmeldy to treat pediatric patients with MLD and enjoys the FDA’s Priority Review status. A decision from the regulatory body is expected on Mar 18, 2024.
MLD is a rare hereditary disease of the body’s metabolic system. In its most severe manifestation, babies develop normally but start to rapidly lose the ability to walk, talk and interact in late infancy.
Including this CVR entitlement, Kyowa Kirin is liable to pay $17 per ADS to Orchard’s shareholders, bringing up the transaction value to approximately $477.6 million, subject to meeting certain conditions.
The transaction has been unanimously approved by the board of directors of both companies and is expected to close in the first quarter of 2024, subject to shareholder approval and the fulfillment of certain regulatory and customary closing conditions.
ORTX’s stock shot up 97.4% on Thursday, in response to the acquisition news. Year to date, shares of the company have skyrocketed 330% against the industry’s 17.8% fall.
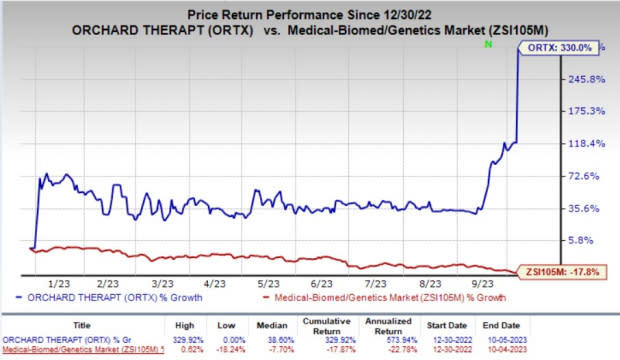
Image Source: Zacks Investment Research
The successful closing of the transaction will provide Kyowa Kirin access to Orchard’s novel gene-therapy approach, whereby it uses a patient’s own genetically modified hematopoietic stem cells (HSCs) to potentially correct the underlying cause of a genetic disease using a single administration.
The transaction will add Orchard’s portfolio of commercial, clinical and pre-clinical HSC gene therapies designed to address serious diseases with significant unmet medical needs to Kyowa Kirin’s portfolio.
ORTX’s commercial portfolio includes only one marketed product, Libmeldy. The addition of Libmeldy to Kyowa Kirin’s portfolio will provide it with an incremental stream of revenues, thereby boosting the top line. Kyowa Kirin is set to accelerate the development of Orchard’s next-in-line MPS programs and its other early-stage programs.