Mirati Therapeutics MRTX reported a loss of $2.49 per share for third-quarter 2023, which was narrower than the Zacks Consensus Estimate of a loss of $2.92 as well as the year-ago quarter’s loss of $3.09 per share.
Mirati reported $16.4 million in revenues for the third quarter, missing the Zacks Consensus Estimate of $19.02 million. Mirati had recorded revenues of $5.4 million in the year-ago quarter.
Quarter in Detail
Third-quarter revenues comprised $16.4 million as product revenues from Krazati/adagrasib, Mirati’s new cancer drug, compared with $13.4 million in the previous quarter. The FDA approved Krazati, a KRAS G12C inhibitor, to treat adult patients with KRASG12C-mutated locally advanced or metastatic non-small cell lung cancer (NSCLC) in December 2022. No product revenues were recorded in the year-ago quarter.
So far this year, the stock price of Mirati has risen 24.5% against the industry’s 21% decline.
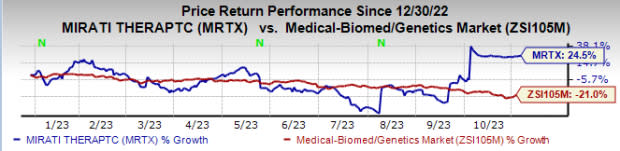
Image Source: Zacks Investment Research
There were no license and collaboration revenues in the quarter compared with $5.4 million in the year-ago quarter.
Research and development expenses declined 12.4% from the prior-year quarter’s level to $114.8 million. This decrease was due to a reduction in clinical development costs for sitravatinib as the company scrapped the development of the candidate, and a decline in share-based compensation.
Selling, general and administrative expenses rose 18.4% from the year-ago quarter’s level to $72.0 million due to an increase in commercial-related costs to support the marketing and sales of Krazati and higher headcount-related costs.
Cash, cash equivalents and short-term investments as of Sep 30, 2023 were $976.4 million compared with $779.4 million as of Jun 30, 2023.
In July 2023, Bristol Myers BMY offered to buy Mirati in an all-cash transaction for $58 per share, aggregating to $4.8 billion. Existing shareholders are also eligible to receive a non-tradeable contingent value right (“CVR”) per share, entitling the holder to receive an additional $12 per share. The CVR adds another $1 billion to the transaction, bringing the deal's total value up to $5.8 billion. The transaction is anticipated to close by the first half of 2024, pending approval of shareholders as well as other regulatory approvals.
Pipeline Update
A regulatory filing for Krazati is under review in Europe. In July, the European Medicine Agency’s Committee for Medicinal Products for Human Use (CHMP) gave a negative opinion regarding the granting of conditional marketing authorization to Krazati. The CHMP mentioned that though the drug has a favorable risk-benefit profile, it does not meet certain requirements for conditional approval. A re-examination of CHMP’s opinion is ongoing.