-
Cash Position: Increased to $229.8 million as of December 31, 2023, from $154.3 million the previous year.
-
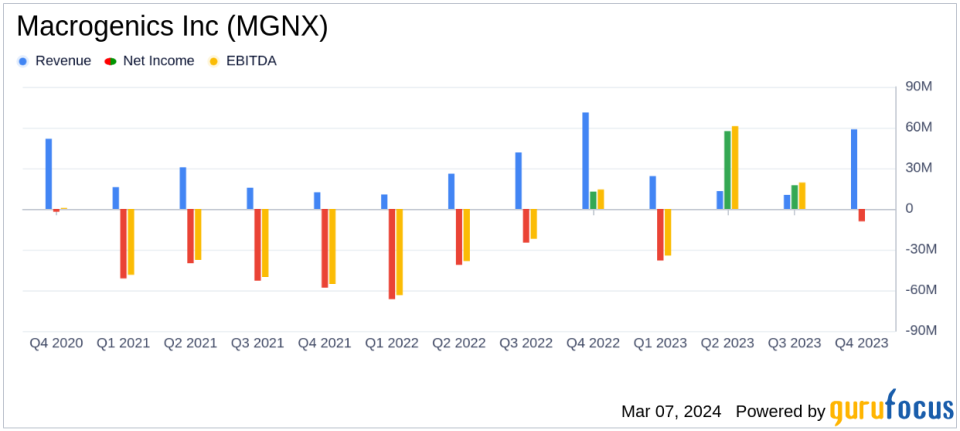
Revenue: Total revenue decreased to $58.7 million in 2023 from $151.9 million in 2022, primarily due to a decrease in collaborative and other agreements.
-
R&D Expenses: Decreased to $166.6 million in 2023 from $207.0 million in 2022, reflecting lower manufacturing and development costs.
-
SG&A Expenses: Decreased to $52.2 million in 2023 from $58.9 million in 2022, mainly due to reduced selling costs for MARGENZA.
-
Net Loss: Significantly reduced to $9.1 million in 2023 from a net loss of $119.8 million in 2022.
-
Shares Outstanding: As of December 31, 2023, there were 62,070,627 shares of common stock outstanding.
On March 7, 2024, Macrogenics Inc (NASDAQ:MGNX) released its 8-K filing, detailing the company's financial results for the year ended December 31, 2023, and providing an update on its corporate progress. Macrogenics, a biopharmaceutical company, is at the forefront of developing antibody-based therapeutics for cancer treatment, including its product MARGENZA for metastatic HER2-positive breast cancer. The company's strategy leverages partnerships to develop a broad portfolio of pipeline candidates, targeting not only cancer but also autoimmune disorders and infectious diseases. Macrogenics' intellectual property is protected by patents on the composition of its product candidates and the technology used to create them.
Financial Highlights and Clinical Advancements
Macrogenics reported a solid cash position of $229.8 million, an increase from the previous year's $154.3 million. This financial strength is expected to extend the company's cash runway into 2026, supporting ongoing clinical and preclinical studies. Despite a decrease in total revenue from the previous year, the company significantly reduced its net loss from $119.8 million in 2022 to $9.1 million in 2023. This improvement was aided by a gain on royalty monetization arrangement and a milestone payment related to TZIELD.
Research and development expenses saw a decrease, reflecting lower costs associated with manufacturing and clinical trials. Selling, general, and administrative expenses also declined, primarily due to decreased selling costs for MARGENZA. The company's commitment to advancing its pipeline is evident in the progress of several key investigational programs, including the TAMARACK Phase 2 study of vobra duo in mCRPC patients, the initiation of a Phase 1 study of MGC026, and the anticipated submission of an IND for MGC028 by year-end.