Karuna Therapeutics, Inc. KRTX reported a loss of $3.01 per share in fourth-quarter 2023, wider than the Zacks Consensus Estimate of a loss of $2.64.
In the year-ago quarter, the company posted a loss of $2.22 per share. The loss increased from the year-ago quarter due to higher operating expenses.
In the fourth quarter of 2023, Karuna did not record any revenues. Sales missed the Zacks Consensus Estimate of $2.9 million. In the year-ago quarter, the company recorded $5.3 million in licensing revenues.
Quarter in Detail
In the reported quarter, research and development expenses were $82.2 million, up 24.5% from the year-ago quarter’s figure, due to increased costs related to lead pipeline candidate KarXT (xanomeline-trospium) plus higher employee-related expenses.
General and administrative expenses surged almost 95% year over year to $47.4 million, driven by pre-commercialization activities for KarXT and higher employee-related costs.
As of Dec 31, 2023, Karuna had cash, cash equivalents and marketable securities of $1.3 billion, similar to what it had as of Sep 30, 2023.
Shares of Karuna have rallied 61.5% in the past year against the industry’s decline of 8.5%.
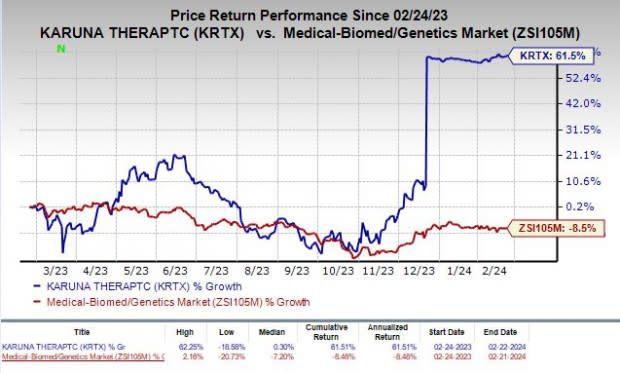
Image Source: Zacks Investment Research
Full-Year Results
In 2023, Karuna generated revenues of $0.6 million compared with $10.6 million generated in 2022.
In the same period, the company recorded a loss per share of $11.73 compared with $8.74 in 2022.
Recent Updates
Karuna is all set to be acquired by drug giant Bristol Myers BMY for a total equity value of $14 billion.
In December 2023, the companies entered into a definitive merger agreement wherein Bristol Myers agreed to acquire all outstanding shares of Karuna for $330 per share in cash.
The transaction, expected to be completed by first-half 2024, is subject to customary closing conditions and clearance from regulatory authorities. Karuna and BMY’s board of directors have already approved this transaction.
An acquisition by Bristol Myers, which has high reserves of cash flow, would enable the company to ramp up the development of its pipeline.
Karuna’s KarXT is an antipsychotic with a novel mechanism of action and differentiated efficacy and safety. The new drug application for KarXT for treating schizophrenia in adults is currently under review in the United States. The FDA has set a target action date of Sep 26, 2024.
If approved, KarXT will provide a new treatment option for patients and be the first novel pharmacological approach for schizophrenia treatment in several decades.
KarXT is being evaluated in the phase III ARISE study as adjunctive treatment in schizophrenia. Top-line data from this study is expected in 2025. Meanwhile, the phase III ADEPT-1 and ADEPT-2 studies are evaluating KarXT in psychosis in Alzheimer’s disease. Top-line data from this study is expected in 2026.