Shares of ORIC Pharmaceuticals, Inc. ORIC tanked 28.7% after the company announced the discontinuation of pipeline candidate ORIC-101 concurrent with fourth-quarter results.
The candidate was being studied in combination with nab-paclitaxel in various solid tumors and in combination with Xtandi in metastatic prostate cancer.
ORIC conducted planned interim analyses of two phase Ib studies and concluded that ORIC-101 did not demonstrate sufficient clinical activity to warrant further development.
The dose-expansion portion of the phase Ib clinical trial of ORIC-101 in combination with Xtandi enrolled 28 patients at the recommended phase II dose (RP2D) with metastatic prostate cancer progressing on enzalutamide. The combination did not translate into a meaningful clinical benefit, with a median progression-free survival (PFS) in the target patient population of 3.7 months.
The dose-expansion portion of the phase Ib study of ORIC-101 in combination with nab-paclitaxel enrolled 61 patients at the RP2D across four cohorts: pancreatic ductal adenocarcinoma (PDAC), ovarian cancer, triple-negative breast cancer (TNBC), and other advanced solid tumors. However, there were no objective responses in the PDAC cohort (n=21) and there was one confirmed partial response in the ovarian cancer cohort (n=13). But no meaningful clinical benefit based on median PFS across all cohorts was observed.
The results disappointed investors.
Consequently, due to the discontinuation of this candidate, ORIC projects its cash runway to extend into the second half of 2024.
Shares of the company have plunged 68.7% in the year so far compared to the industry’s fall of 12.2%.
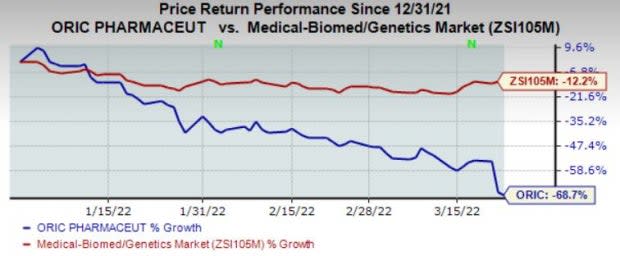
Image Source: Zacks Investment Research
Other candidates in the pipeline include ORIC-533: CD73 Inhibitor, ORIC-114: EGFR/HER2 inhibitor, and ORIC-944: PRC2 inhibitor.
A phase Ib trial with ORIC-533 as a single agent in multiple myeloma is enrolling patients and initial data are expected to be reported in the first half of 2023.
A phase Ib trial with ORIC-114 as a single agent has been initiated and will enroll patients with advanced solid tumors with EGFR or HER2 exon 20 alterations or HER2 amplification and will allow patients with CNS metastases that are either treated or untreated but asymptomatic. The company expects to report initial Phase 1b data from this trial in the first half of 2023.
A phase Ib trial with ORIC-944 as a single agent will enroll patients with metastatic prostate cancer. The company expects to report initial Phase 1b data from this trial in the first half of 2023.