Immuneering Corporation IMRX is a clinical-stage biotech company focused on developing universal-RAS/RAF medicines to treat cancer patients.
Devoid of marketed drugs in its portfolio, Immuneering is entirely dependent on its pipeline for growth. The company’s lead pipeline candidate is IMM-1-104, an investigational RAS medication, which is being evaluated in a phase I/IIa study in patients with advanced solid tumors harboring RAS mutations.
In the past week, Immuneering’s shares have plunged 70.9% compared with the industry’s 2.1% fall. This downside came after the company reported topline results from the phase I portion of the phase I/IIa study on IMM-1-104 last week.
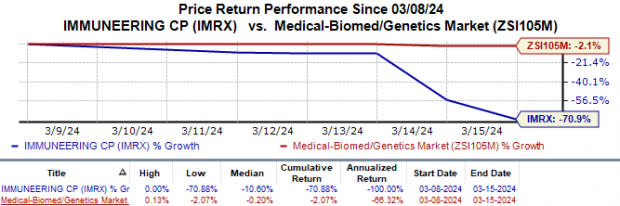
Image Source: Zacks Investment Research
The endpoints of the phase I portion of the study were to assess the safety and tolerability of IMM-1-104, identify a candidate recommended Phase 2 dose (RP2D), and evaluate pharmacokinetics (PK). As of Feb 20 (cut-off date), management enrolled heavily-pretreated study participants who had a dozen different RAS mutations across eight different types of cancer.
As of the cut-off date, treatment with the drug was well-tolerated among study participants, with the occurrence of just one grade 3 event (non-serious rash). Results from the study demonstrated clear proof of concept, as treatment with IMM-1-104 showed promising signs of initial clinical activity through deep cyclic mechanisms. Based on this data, management decided to proceed with a 320mg dose of the drug as RP2D.
The above results failed to impress investors. Though the data does warrant clinical advancement of Immuneering’s lead drug, a lack of confirmed responses observed across different doses (including the RP2D) did not sit well with investors. With the study participant population being very diverse and low in number (n=41), it was difficult to point out the level of clinical activity in patients treated with the drug with clarity.
Due to the above factors, multiple Wall Street analysts downgraded the IMRX stock. Analysts at Jeffries downgraded the stock from ‘Buy’ to ‘Hold’ and also set the price target to $3 per share from the previously set target of $16.
Prior to the announcement of the above results, management started dosing patients in the phase IIa portion of the study. This portion will evaluate a 320mg dose of IMM-1-104 as monotherapy in pancreatic ductal adenocarcinoma (PDAC), non-small cell lung cancer (NSCLC) and melanoma, and as combination therapy in PDAC.
Initial data from the phase IIa portion is expected later this year. The results from this portion are expected to provide a clearer picture as management intends to enroll more study participants at a longer follow-up period.