IGM Biosciences IGMS, a clinical-stage company, is developing a pipeline of Immunoglobin M (IgM) antibody-based candidates to treat patients with cancer and autoimmune and inflammatory diseases.
The company believes that an IgM has the potential to become a better antibody medicine, given the limitations of the conventionally used IgG antibody.
Over the past three months, shares of IGM Bio skyrocketed 142% compared with the industry’s 10.4% rise. The primary reason behind the massive uptick in the stock price was the company’s strategic pipeline reprioritization efforts announced in December 2023. Investors cheered the company’s decision to streamline its pipeline to ensure efficient utilization of resources.
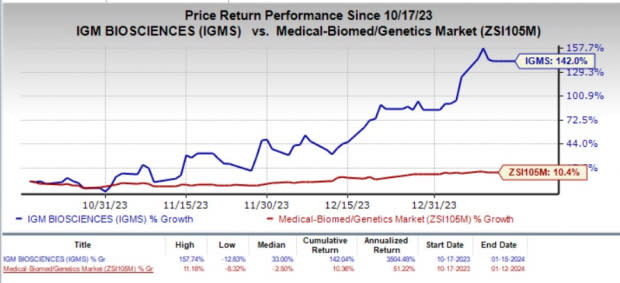
Image Source: Zacks Investment Research
As part of its reprioritization efforts, IGM Bio said that it will focus its resources on two strategic areas, such as treating colorectal cancer using IgM death receptor 5 (DR5) agonist antibodies and autoimmune diseases using IgM T cell engager antibodies.
IGMS is halting all hematologic oncology clinical development as well as the clinical development of its targeted cytokine product candidate. The company will also be cutting its workforce by approximately 22%.
Such measures, taken by IGM Bio, are expected to extend its cash runway into the second quarter of 2026.
The company’s oncology pipeline comprises aplitabart, a DR5 agonist. An early-stage study is currently enrolling patients to evaluate aplitabart in combination with Folfiri chemotherapy and bevacizumab, a monoclonal antibody, for the treatment of second-line metastatic colorectal cancer. IGM Bio expects to enroll approximately 110 patients by the end of the first quarter of 2024.
IGMS also continues to enroll later-line colorectal cancer patients in its combination clinical study of 10 mg/kg of aplitabart and Folfiri. Enrollment in this combination study is expected to be completed in the first half of 2024.
IGM Bio is also currently developing imvotamab, an IgM-based antibody, in autoimmune diseases. Two separate phase Ib studies are ongoing and evaluating the candidate in severe systemic lupus erythematosus and severe rheumatoid arthritis.
The company also recently received FDA clearance for its investigation new drug (IND) application for the use of imvotamab in treating idiopathic inflammatory myopathies. IGMS is currently preparing to initiate an early-stage study on imvotamab for this additional indication.
Besides imvotamab, IGM Bio has another IgM-based antibody intended for clinical development in autoimmune diseases, IGM-2644, which is currently in the pre-clinical stage. The company plans to file an INDapplication for IGM-2644 in 2024 to begin clinical development for autoimmune indications.