Crinetics Pharmaceuticals’ CRNX shares gained 19.1% on Mar 19 after the company reported positive top-line data from the second (PATHFNDR-2) of two late-stage studies evaluating the efficacy and safety of lead candidate, paltusotine, for the treatment of acromegaly.
Acromegaly is a rare endocrine disease generally caused by a pituitary adenoma, which is a non-cancerous tumor in the pituitary that secretes growth hormone.
The PATHFNDR-2 study is evaluating paltusotine, an oral SST2 agonist, in participants with acromegaly who are treatment-naïve or not currently receiving medical therapy.
Per the data readout, the phase III PATHFNDR-2 study met its primary endpoint, with statistical significance, observing that 56% of patients treated with paltusotine achieved an insulin-like growth factor 1 (IGF-1) level ≤ 1.0 times the upper limit of normal (xULN) compared with those who received placebo (5%). Crinetics also reported that all secondary endpoints met statistical significance.
Additionally, CRNX stated that the candidate was overall well tolerated with no serious adverse events in the PATHFNDR-2 study.
Management believes that the encouraging data from the PATHFNDR-2 study reaffirms paltusotine’s potential to provide both symptom control as well as biochemical control in patients who are not currently on pharmacologic treatment for acromegaly.
In the past year, shares of Crinetics have skyrocketed 179.4% against the industry’s 5% decline.
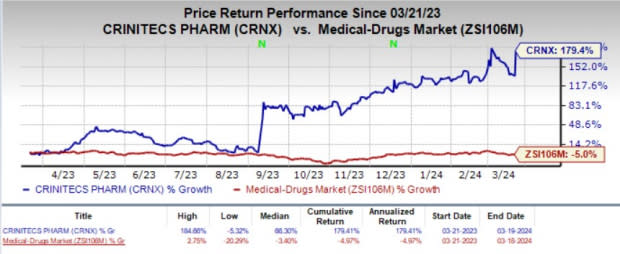
Image Source: Zacks Investment Research
We would like to remind the investors that in September 2023, CRNX reported positive top-line data from the first late-stage study of paltusotine for the treatment of acromegaly, PATHFNDR-1.
The phase III PATHFNDR-1 study evaluated paltusotine in patients with acromegaly who were switching from current standard-of-care treatment, octreotide or lanreotide depot monotherapy.
Per the data readout, the study met its primary endpoint with statistical significance, observing that 83% of participants taking paltusotine maintained an IGF-1 level ≤ 1.0xULN compared with those who received placebo (4%). All secondary endpoints in the phase III PATHFNDR-1 study also met statistical significance.
Positive data readout from both the phase III PATHFNDR-1 and PATHFNDR-2 studies bolsters paltusotine’s potential in becoming a new treatment option for all patients living with acromegaly, subject to approval.
Crinetics is currently gearing up to submit a regulatory application to the FDA seeking approval of paltusotine for the acromegaly indication in the second half of 2024. The company aims to potentially launch the product in the U.S. market in 2025.