Acadia Pharmaceuticals Inc. ACAD shares lost 15% in the after-market hours on Mar 11, after the company announced the failure of its late-stage study evaluating pimavanserin for the treatment of negative symptoms of schizophrenia.
In the phase III ADVANCE-2 study, treatment with pimavanserin did not achieve statistical significance in demonstrating improvement over placebo on the study’s primary endpoint of change from baseline to week 26 on the Negative Symptom Assessment-16 (NSA-16) total score. The NSA-16 scale is a widely used metric to measure change in the wide range of predominant negative symptoms that patients experience.
Based on such disappointing results, Acadia has decided not to pursue any further clinical studies with pimavanserin. However, the company will continue to analyze data from the schizophrenia study of pimavanserin.
Pimavanserin was, however, well-tolerated in the ADVANCE-2 study and had a safety profile consistent with previous clinical studies. Adverse events related to treatment with the drug were minimal.
In the past year, shares of Acadia have gained 24.3% against the industry’s 4.6% decline.
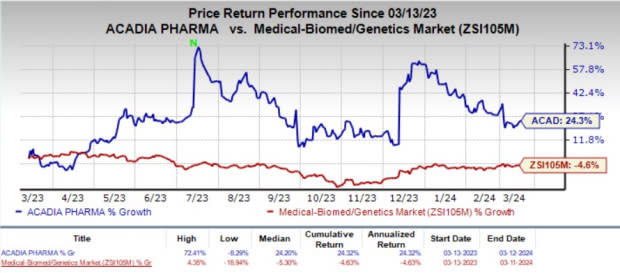
Image Source: Zacks Investment Research
Notably, ACAD is not new to developmental/regulatory setbacks for pimavanserin. The company had previously failed to gain label expansion for pimavanserin to treat hallucinations and delusions associated with dementia-related psychosis and Alzheimer’s disease psychosis (ADP) in 2021 and 2022, respectively.
We remind the investors that Acadia’s pimavanserin is currently marketed in the United States as the first and only FDA-approved treatment for hallucinations and delusions associated with Parkinson’s disease psychosis. The drug was launched in May 2016 under the brand name Nuplazid.
Nuplazid recorded sales worth $549.2 million in 2023, up 6% year over year. Total Nuplazid sales accounted for about 76% of the company’s revenues in 2023.
Despite the significant setback leading to the abandonment of further clinical studies with pimavanserin, Acadia’s pipeline beyond Nuplazid is reassuring.
The company is currently evaluating the efficacy and safety of carbetocin nasal spray (ACP-101) in a phase III COMPASS study for treating hyperphagia associated with Prader-Willi Syndrome.
The company is also developing ACP-204 as a potential treatment for ADP. ACAD completed phase I development of ACP-204, witnessing a favorable safety and tolerability profile. In the fourth quarter of 2023, Acadia announced that it has initiated a mid-stage study to evaluate the safety and efficacy of ACP-204 to treat hallucinations and delusions associated with ADP.
The phase II study, initiated by the company, is the first part of the planned phase II/III clinical program for ACP-204 in the treatment of ADP. The mid-late-stage program comprises a single phase II study and two phase III studies that have almost identical designs.