Candel Therapeutics, Inc. CADL announced that the FDA has granted Fast Track designation to its investigational adenovirus candidate CAN-2409 plus prodrug (valacyclovir) for the treatment of pancreatic ductal adenocarcinoma (PDAC). Shares of the company were up 20.6% on Dec 12 following the announcement of the news.
The Fast Track designation facilitates rapid development and expedites the review of drug candidates that are being developed to treat serious conditions and for which clinical data demonstrate the potential to address unmet medical needs. The goal is to make these treatments rapidly available to patients in need.
CAN-2409 is currently being evaluated in a phase II study for treating patients with PDAC and improve overall survival.
In November 2023, based on an interim analysis, CADL reported encouraging overall survival and immunological biomarker data from the phase II study evaluating CAN-2409 plus prodrug with standard of care — neoadjuvant chemoradiation followed by resection in borderline resectable non-metastatic PDAC.
Data from the same study demonstrated prolonged and sustained survival after experimental treatment with CAN-2409 in the given patient population.
Candel plans to announce updated overall survival data from the interim analysis of the above-mentioned study in the second quarter of 2024.
Shares of Candel have plunged 42.6% in the past year compared with the industry’s decline of 23.7%.
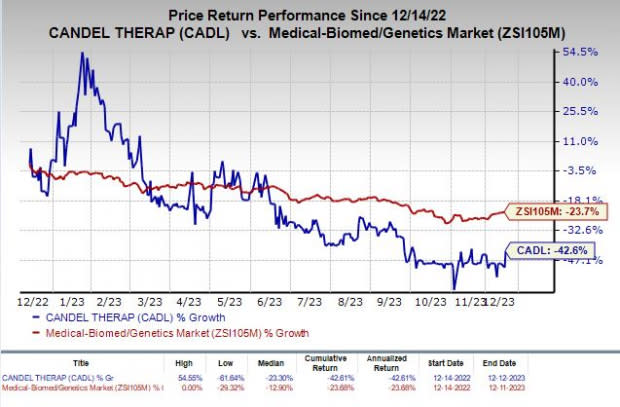
Image Source: Zacks Investment Research
Last month, CADL announced strategic restructuring and portfolio prioritization with a focus on its lead asset, CAN-2409, and the clinical development of another candidate, CAN-3110.
As part of the restructuring, the company reduced its current workforce by almost 50%, which is likely to reduce costs and operating expenses. This is likely to extend Candel’s cash runway into the fourth quarter of 2024.
The company expects key data readouts for CAN-2409 in non-small cell lung cancer and pancreatic cancer in the second quarter of 2024, while top-line data in prostate cancer is expected in the fourth quarter of 2024.
Candel is also advancing the development of CAN-3110 in recurrent high-grade glioma. Initial biomarker data on CAN-3110 is expected in the second half of 2024.
In the absence of a marketed product, the successful development of its pipeline candidates remains the key focus for this clinical-stage biopharmaceutical company.
Zacks Rank & Stocks to Consider
Candel currently carries a Zacks Rank #4 (Sell).
Some better-ranked stocks in the biotech sector are Journey Medical Corporation DERM, Entrada Therapeutics, Inc. TRDA and Puma Biotechnology, Inc. PBYI, each sporting a Zacks Rank #1 (Strong Buy). You can see the complete list of today’s Zacks #1 Rank stocks here.